Huge Recall of Children’s Ibuprofen, Apotex Eye Drops, Dangerous Dietary Supplements; Warnings for Levodopa, Xeljanz, Migraine Drugs, Sivextro, Children’s Xyzal; Antibiotics Research, Cancer Research
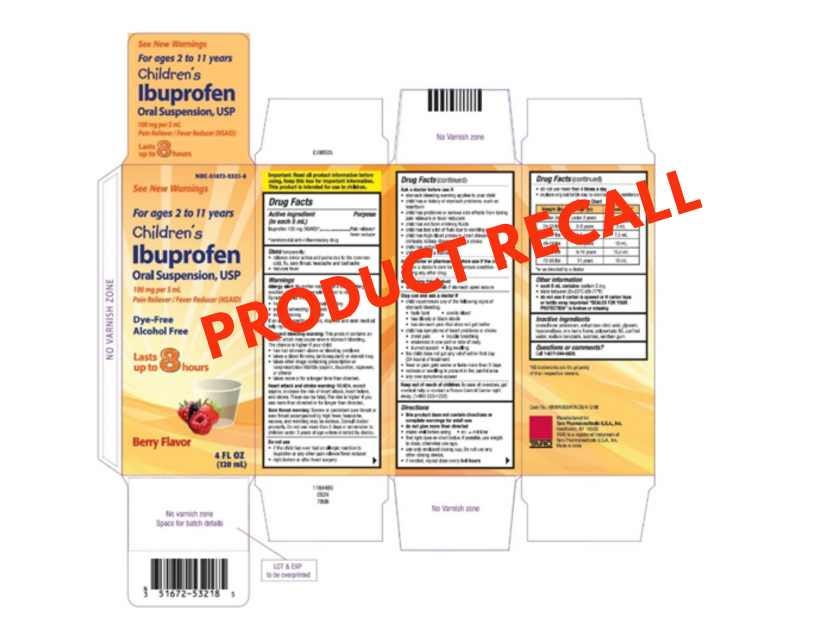
Children’s Ibuprofen Recall (Taro Pharma)
Strides Pharma is recalling almost 90,000 bottles of over-the-counter Children’s Ibuprofen liquid formulation due to a consumer finding a gel-like mass and black particles in the product. The medication was made in India for Taro Pharma USA. Check this Enforcement Report for lot numbers and expiration dates.
Glaucoma Eye Drops Recall by Apotex
More than 107,00 vials (5mL) of Brimonidine Tartrate/Timolol Maleate Ophthalmic Solution because of the lack of assurance of sterility. This is for the 0.2%/0.5% strength medication, and the severity of the recall has not yet been classified by the FDA. Apotex 2026 Recall
In October 2025, Apotex had another large recall (still ongoing) of over 150,000 eye drop bottles of brimonidine tartrate/timolol maleate along with 493,000 bottles of prescription Ketorolac Tromethamine eye drops for allergies.
Ask a Patient News: Eye Drops by Apotex Recalls Fall 2025

Two Dietary Supplements Recalled Nationwide
Anthony Trinh, 123Herbals LLC is recalling 25-count bottles of Silintan capsules, which it sells as a dietary supplement for pain relief. FDA detected that the product contains undeclared meloxicam, a prescription nonsteroidal anti-inflammatory drug (NSAID) for arthritis. This medication can have serious side effects and drug interactions. Read the FDA Enforcement Report and FDA Press Release for Consumers for more details.
Also, a dietary supplement marketed for male enhancement, MR. 7 SUPER 700000 Capsules, sold online by StuffbyNainax, is under a Class I recall. The FDA detected that the product contains undeclared sildenafil and tadalafil, prescription-only ingredients used to treat erectile dysfunction. Read the FDA Press Release for Consumers and FDA Enforcement Report for more details, including the serious risks associated with taking these supplements.
Drug Warning: Parkinson’s Drug and Seizures from Vitamin Deficiency
The FDA issued a Drug Safety Communication regarding Parkinson’s medications containing carbidopa/levodopa, stating that after a safety review, it determined that the drugs can deplete vitamin B6 levels, resulting in a vitamin deficiency that can lead to seizures. Products that will soon carry a new warning about the risk include carbidopa/levodopa, Rytary, Sinemet, Sinemet CR; carbidopa/levodopa/entacapone (Stalevo), foscarbidopa/foslevodopa (Vyalev). These products include multiple formulations and may be administered by several different routes, including oral tablets, an enteral (intestinal) suspension, and a subcutaneous injection.
FDA Safety Communication: “FDA Requiring Warning About Vitamin B Deficiency and Associated Seizures in Drug Products Containing Carbidopa/Levodopa”
Xeljanz and Risk of Infections, Fractures
Rheumatoid Arthritis (RA) drug Xeljanz (tofacitinib) expanded warnings about the serious risk of infection. It also added new data from a clinical trial safety study of adults with Rheumatoid Arthritis that tested higher doses of the drug (10 mg twice a day) compared with lower doses (5 mg twice a day) and TNF blockers. Xeljanz, and Xeljanz higher dose were associated with a higher risk of fractures than the TNF blocker. Given these increased risks, Xeljanz (tablets and oral solution) 10 mg twice daily or XELJANZ XR (extended-release tablets 22 mg once daily) dosages are not recommended for the treatment of Rheumatoid Arthritis (RA), Psoriatic Arthritis (PsA), Ankylosing spondylitis (AS), or polyarticular course juvenile idiopathic arthritis (pcJIA).
Topamax Migraine Drug and Serious Skin Reactions
Migraine and seizure drug Topamax (topiramate), along with other topiramate-containing drugs, have new warnings that severe hypersensitivity reactions, including anaphylaxis and skin swelling. Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as multiorgan hypersensitivity, has also been reported in patients taking topiramate. DRESS may be fatal or life-threatening.
At the FDA’s request, the labels for Topamax, Eprontia, and Trokendi XR were updated to warn about these risks. Topamax is approved to treat epilepsy/seizures in patients age 2 and up and as a preventive migraine treatment for age 12 and up. It was originally approved in 1996.
Ozurdex (dexamethasone) Intravitreal (Eye) Implant Warning
Ozurdex is an eye implant that releases the drug dexamethasone (a steroid) to treat three serious eye conditions: diabetic macular edema (complication of diabetes that can cause blindness), non-infectious uveitis (inflammation in central section of the eye), and central retinal vein occlusion, which is blockage of the small veins that carry blood away from the retina.
Ozurdex drug label was updated to warn about the risk of “corneal decompensation” that could happen if the device is dislocated. Corneal decompensation happens when the cornea loses its ability to maintain proper hydration and clarity, and causes swelling and cloudiness. This can lead to vision problems and may require additional treatment. Instructions for device insertion were revised. After implanting the device, patients should be monitored for elevation of eye pressure and for endophthalmitis, a serious eye infection. Ozurdex was approved in 2009.
Read more about uveitis at Mayo Clinic: Uveitis Symptoms and Causes
Read more about diabetic macular edema at Mayo Clinic: Screening for diabetic macular edema
Read more about retinal vein occlusion at Medline Plus.
Children’s Xyzal (levocetirizine) and Severe Withdrawal Itching Side Effect
In a letter, FDA approved the revision to Children’s Xyzal Allergy 24 Hour oral solution over-the-counter packaging to add a warning about the risk of extreme itching (“new onset pruritis”) to be added to the product’s “Drug Facts.” The FDA requested this change in July 2025 and recognized the side effect, which is a type of withdrawal effect associated with long-term use of prescription and over-the-counter allergy medicines cetirizine (Zyrtec) and levocetirizine (Xyzal) in a Drug Safety Communication in May 2025. For years, anecdotal reports of the withdrawal itching side effect have been received through sites like Ask a Patient and People’s Pharmacy and through the FDA’s adverse events reporting system.
Prescription products had the new warnings first, beginning last year: Xyzal levocetirizine dihydrochloride tablets and withdrawal itching and “Beware of Itching After Discontinuing These Allergy Drugs”
Sivextro Antibiotic and Serotonin Syndrome
Antibiotic Sivextro (tedizolid phosphate) added a warning about the risk of serotonin syndrome when Sivextro is taken with certain drugs. Serotonin syndrome is a potentially life-threatening condition with symptoms including fever, sweating, agitation, unusual eye movements, difficulty with coordination, stiff muscles, and twitching.
Drugs (serotonergic agents) include antidepressants, prescription opioid pain medications, anxiety medications, methotrexate, topotecan, rosuvastatin, and some migraine headache medications. Patients taking these drugs along with Sivextro should be monitored for signs and symptoms of serotonin syndrome.
The label was also updated with the side effect of Clostridioides difficile-associated diarrhea and replaced the pediatric age specification for tablets (age 12) with a weight specification of at least 77 pounds (35 kg). Sivextro is administered by I.V. or in tablet form and treats skin and skin structure infections in children and adults. It was approved in 2014.
The antibiotic Linezolid (Zyvox brand name) is a similar antibiotic to Sivextro – both are synthetic “oxazolidinone” antimicrobial agents. FDA issued a drug safety communication related to Zyvox in 2011: Risk of Serotonin Syndrome and Linezolid Zyvox
Antibiotics Research: Azythromycin Not Helpful for Covid
University of California San Francisco researchers found that the routine use of the antibiotic azithromycin (Zithromax brand name) in hospitalized patients during the COVID-19 pandemic had no clinical benefit and was associated with potentially harmful changes in the upper respiratory microbiome. The researchers found an increase in the expression of antibiotic-resistant genes after just one day of exposure to the antibiotic. Alarmingly, it was found was that patients who received azithromycin had a reduced presence of some harmless bacteria and an increased presence of pathogenic species, such as Staphylococcus and Klebsiella, and more “detectably expressed,” or active, macrolide resistance genes in their upper airway. The study was published in Nature Microbiology.
CIDRAP: “Antibiotic used in COVID patients tied to increased signs of antibiotic resistance”
Antibiotics Research: Effects on the Gut Microbiome Can Last for Many Years
Researchers at Uppsala University, Karolinska Institutet, and Lund University studied how antibiotic use affects the gut microbiome over time. The study, based on drug registry data and genetic analysis of fecal samples from nearly 15,000 Swedish residents, found that people who took certain oral antibiotics had fewer bacterial species in their gut, and a lower abundance of individual species of gut bacteria, for four to eight years after taking them.
The antibiotics most strongly associated with these long-lasting effects were clindamycin, fluoroquinolones, and flucloxacillin. Some patients can have problems with their gut, such unexplained diarrhea, irritable bowel, or inflammatory bowel conditions, for many years after just taking one course of antibiotics. The findings were published in Nature Medicine.
CIDRAP: “Antibiotic use linked to ‘persistent’ gut microbiome changes”
Related: Dr. Lucy McBride just answered a question on Friday from a reader who likely read this same research - Should a patient be worried about long-term disruption to the gut microbiome if she gets a new antibiotic prescription for a sinus infection?
Metformin Potential for Cancer Prevention
Type 2 diabetes drug metformin was first approved under the name Glucophage in Europe in the 1950s and is emerging as a “wonder drug.” It has been studied for treating Long Covid, improving longevity, lowering dementia risk for those with type 2, as a treatment for kidney disease, for lung conditions like COPD, and more. Joe Graedon of People’s Pharmacy recently updated his article on metformin and cancer.
He says that while there is conflicting data for whether taking metformin makes any difference for prostate cancer, recent research in JAMA Network Open, March 16, 2026 has encouraging news about metformin and esophageal cancer. The scientists report that people taking metformin had a 36% lower likelihood of being diagnosed with esophageal squamous cell carcinoma than those who were not taking it. Higher doses were associated with even lower risk, about 48%.
The People’s Pharmacy: “Should Everyone Be Taking Metformin to Fight Cancer?”
New Medications Roundup: Wegovy HD, Leucovorin, Plaque Psoriasis Pill, Meds for Liver-Related Itching, Growth Drug for Kids with Achondroplasia
Check out the key facts about recent new FDA drug approvals and expanded drug approvals at our website.
AskaPatient.com: “Recent Drug Approvals: Wegovy HD, Leucovorin, Plaque Psoriasis, Liver-Related Itching Meds, More”
Did you miss our last newsletters from March 8? Check them out here:






Metformin a wonder drug? good grief. 3 years ago, I had to find a new doc as the old one retired. This woman prescribed it to me. I took only 4 tablets of it and stopped, because of heavy stomach cramps, heavy black diarrhea, unable to eat (even though I had to take it with food), then my arms and legs started to cramp up - thankfully in a lucid moment I looked this up and found, that going on with it could cause cramps in vital organs and death. The article stated, that 45% of patients have slight to moderate side effects of the thing! No need to say I never went back to that doc and found another one, who said I am not diabetic.
Anything and everything produced by Big Pharma is now dangerous. Not that they have ever been safe before...