April 2026 Part 2: Recalls for Clonidine and Xanax XR; Health fraud scams; New warnings for Proscar, Eylea; Foundayo safety; Medication shortages; Lung cancer surgery, more
Clonidine Patch Recall by Teva
Teva is recalling 124,054 cartons of high blood pressure treatment clonidine transdermal system (patch), .1 mg/day dosage, 62,136 cartons at the .2 mg dosage, and 113,943 cartons of the .3 mg strength dosage due to use of an unapproved raw material during manufacturing. The patches, packed four to a carton, were manufactured by Actavis in Salt Lake City, Utah and distributed by Actavis nationwide. Clonidine (Catapres TTS brand name) patch is applied to the skin weekly. While the recall is directed at retailers, consumers may check the recall report for more information.
FDA Recall Report
Xanax XR (alprazolam) Extended-Release Tablets Recall by Viatris
Viatris, Inc. is recalling one lot of 3 mg strength, 60-tablet bottles of panic disorder treatment Xanax (alprazolam) extended-release tablets because the tablets may not dissolve properly (failed dissolution specifications). The tablets were made by Viatris Specialty in Ireland and distributed nationwide by Viatris Specialty in Morgantown, West Virginia. (Lot# 8177156 with an Expiration Date of 02/28/2027)
FDA Recall Report
Additional recent recalls for private label cough drops, isotretinoin, and prazosin are listed at MedShadow Foundation’s recall roundup.
Health Fraud Scam: Arthritis and Pain Relief Supplements with Hyaluronic Acid
FDA is warning consumers not to purchase or use tablets or capsules labeled as containing hyaluronic acid, which are promoted and sold as supplements for arthritis pain relief on websites and possibly in some retail stores. Laboratory testing confirmed the products contain hidden drug ingredients not listed on the product labels. Note that these products are distinct from the hyaluronic acid-containing creams and serums marketed for skin care.
FDA Alert: “Hyaluronic-acid-products-containing-undeclared-ingredients”

Health Fraud Scam: Addall XR Shot and Capsules
FDA is warning consumers not to buy or use Addall XR Shot or Capsules, marketed as dietary supplements to improve focus, using a confusing brand name similar to the FDA-approved ADHD drug Adderall. Laboratory testing found that the orange-flavored Addall XR Shot contains Phenibut (β-phenyl-GABA), an unlawful and dangerous ingredient in dietary supplements, as well as undeclared 1,4-DMAA (1,4-dimethylamylamine). The Addall XL 30 capsules contain the undeclared and unlawful ingredients 2-amino-6-methylheptane (DMHA) and 1,4-DMAA. Phenibut can cause fatigue, balance problems and potential addiction. DMAA and DMHA may cause cardiovascular problems such as high blood pressure or even heart attack. The products are sold online and at retailers such as gas stations and convenience stores nationwide.
FDA Alert: “FDA-advises-consumers-retailers-and-distributors-not-eat-sell-or-distribute-addall-xr-shot-or-addall”

Proscar Warning About Reports of Suicidal Thoughts
Proscar (finasteride), a treatment for benign prostatic hyperplasia (enlarged prostate), added a warning to its label about the risk of suicidal ideation, or thoughts of self-harm or suicide. The medication guide advises that patients taking Proscar should tell their healthcare provider right away if they notice new or worsening depression, mood changes, or suicidal thoughts. Proscar tablets were approved in 1992.
Hair loss drug Propecia, which also contains finasteride, has far more reports of suicidal ideation than does Proscar; a warning was added to its label in 2022. The Ask a Patient database of medication ratings and reviews contains 90 reports of depression or suicidal thoughts associated with Propecia.
Reports of anxiety with Proscar are common; anxiety is the 7th most frequently reported adverse event (400 reports for the side effect out of about 3000) for Proscar in the FDA adverse events database.
Eylea Long-Term Increased Incidence of Stroke and Heart Attack
Eylea HD (aflibercept), a high dose (8 mg) treatment approved in 2023 for four eye diseases including wet age-related macular degeneration, updated its label to add clinical trials results for a long-term (96 weeks) study comparing it with regular 2 mg Eylea. The drug is administered directly into the eye (intravitreal injection) by a healthcare professional.
The trial results showed an increase in the incidence of thromboembolic events (ATEs) over time, but the HD 8 mg formulation, which is given less frequently (every 12 or 16 weeks), had less incidence of the events than regular Eylea 2 mg formulation, which is administered every 8 weeks. ATEs are defined as nonfatal stroke, nonfatal myocardial infarction (heart attack), or vascular death (including deaths of unknown cause). The incidence of ATEs experienced during clinical trials depended on the indication being treated. The following provides the incidence levels by the type of disease being treated:
Wet AMD: 1.8% (12 out of 673 patients) for Eylea HD vs. 3.3% (11 out of 336) for Eylea 2mg
Diabetic Macular Edema: 6.7% (33 out of 491) for Eylea HD vs 7.2% (12 out of 167) for Eylea 2mg
Macular Edema Following Retinal Vein Occlusion through week 36 (a different study): 0.5% (3 out of 591) for Eylea HD vs. 1.7% (5 out of 301) for Eylea 2 mg
The revised label for Eylea HD has additional clinical trial results information.
FDA Eylea HD Safety Update
Read more about age-related macular degeneration (dry and wet), and diabetic retinopathy at the National Eye Institute.
2025 Vulnerable Medicines List and Potential Shortages
The U.S. Pharmacopeia (USP) is a quality standards organization for medicines, dietary supplements, and excipients (inactive ingredients that help deliver the active ingredients in a medicine). USP recently published its annual list of 100 Vulnerable Medicines, a list of clinically important medicines with the most supply chain vulnerabilities that may lead to shortages in the U.S. “Supply chain vulnerabilities are particularly acute when production of key inputs is geographically concentrated or limited to a small number of suppliers,” notes the report.
The majority of at-risk drugs (63%) are injectables, which are always prone to shortages due to their unique production challenges. Oral solid drugs, such as tablets and capsules, represent the second-largest at-risk group at 22%.
Tablets and capsules on the list include at least six that are currently in shortage (noted below with a link to the current shortage report):
Morphine Tablets, Oseltamivir Capsules, Oxycodone Hydrochloride Immediate-Release Tablets (currently in shortage) Isoniazid, Irbesartan, Lisdexamfetamine (ADHD drug in shortage), Amphetamine Aspartate, Amphetamine Sulfate, Dextroamphetamine Saccharate and Dextroamphetamine Sulfate Tablets, Amphetamine Mixed Salts Tablets, Extended-Release Clonazepam Tablets (seizure drug now currently in shortage) Disulfiram Tablets, (used for alcohol addiction), Oseltamivir Capsules and Oral suspension (generic for Tamiflu and in shortage), Methylphenidate immediate and extended release (in shortage) and leucorvorin calcium tablets (in shortage).
View the complete USP vulnerable-medicines-list-2025 (11-page PDF download)
Biospace: “USP-adds-tamiflu-trulicity-to-vulnerable-list-as-upstream-analysis-reshapes-supply-concerns”
Drug Sponsors Reminded to Disclose Clinical Trial Results
The FDA sent notices to more than 2,200 drug and medical product sponsors and researchers (associated with more than 3,000 registered clinical trials, including some that were publicly funded) reminding them of their obligation to submit clinical trial results information to ClinicalTrials.gov, a public database run by the National Institutes of Health’s National Library of Medicine. Trial sponsors, with few exceptions, are required to submit clinical trial results information to ClinicalTrials.gov within one year after trial completion.
According to an internal analysis, FDA says that about 30% of studies that are highly likely to fall under mandatory reporting requirements have not reported any results to ClinicalTrials.gov.
“Far too often, companies are suppressing unfavorable clinical trial results and keeping them secret from patients and the scientific community. Those sponsoring clinical trials have an ethical obligation to make results public regardless of the data’s influence on the company’s share price,” said FDA Commissioner Marty Makary.
What comes next if the companies or researchers don’t send in the trials results? The FDA may send Pre-Notices of Noncompliance and Notices of Noncompliance, and may impose fees as part of risk-based compliance efforts. However, in practice the FDA has rarely sent Notices of Noncompliance or levied fees. The messages recently sent to the trial sponsors, which were not made public, “represent an extra step the agency is taking to provide the relevant responsible parties with the opportunity to comply with federal law before the agency considers whether to take further regulatory action.”
FDA Press Release: FDA-reminds-more-2200-sponsors-and-researchers-to disclose-trial-results
Fierce Biotech: FDA -urges-clinical-trial-sponsors-report-results-their-studies
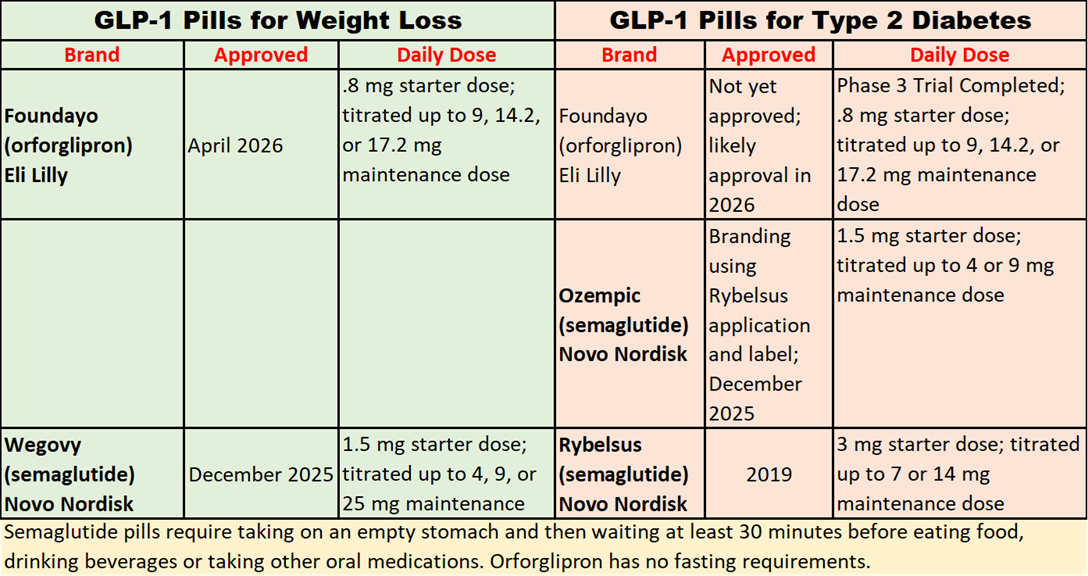
Foundayo’s Rapid Approval and Safety Concerns
Eli Lilly’s new GLP-1 pill for weight loss, Foundayo (orforglipron) was approved on April 1, in a record 50 days under the Commissioner’s National Priority Review pilot program, but FDA’s safety concerns are evident based on the number of required post-approval clinical trials.
When Lilly’s Mounjaro (tirzepatide) GLP-1 medication was approved in 2022, only three post-approval trials were mandated: the standard pediatric trial, a registry-based monitoring study for thyroid cancer (all GLP-1 drugs carry a black box warning for the risk of thyroid tumors), and a lactation study to assess concentrations of tirzepatide in breast milk.
In contrast, the Foundayo (orforglipron) approval letter identifies nine required post-approval studies, including the three that were required of Mounjaro, plus two additional pediatric studies, a pregnancy complications study, a pregnancy exposure registry, a heart attack and stroke and liver injury study, and a delayed gastric emptying study that uses ultrasound to monitor the effects of fasting on stomach contents. Favorable results for the cardiac and liver injury trial will help pave the way for Foundayo’s approval for a Type 2 diabetes indication.
New Favorable Safety Data for Foundayo as Diabetes Treatment
Biospace reported that Lilly’s newly released diabetes data comparing insulin vs. Foundayo showed that the risk of all-cause death to be 57% lower for participants receiving Foundayo and showed 16% lower A1C levels compared to insulin.
Also, the 52-week trial showed “no signs of drug-induced liver injury (DILI), which should assuage FDA concerns about unexpected cases of major adverse cardiovascular events and liver injury.”
Biospace: “Lilly’s Foundayo posts Outstanding Diabetes Data Possibly Easing FDA Safety Concerns”
Weight Loss Results Study and Side Effects
A Novo Nordisk-sponsored study presented at the Obesity Medicine Association’s annual conference showed that Foundayo (36 mg) was associated with around four to 14 times higher odds of treatment discontinuation due to adverse effects compared with semaglutide (Wegovy, 25 mg). It also suggested that the maximum strength Wegovy produced greater average weight loss compared with Foundayo.
Healio: “Obesity Medicine Annual Conference: Semaglutide tied to greater weight loss vs orforglipron”
An included perspective on the study from Fatima Cody Stanford, Healio Primary Care Peer Perspective Board Member suggests that because this is an indirect study, not a head-to-head comparison, results should be interpreted with caution.
An Eli Lilly sponsored study (published in the Lancet) and reported in our March 8 newsletter, showed greater weight loss with orforglipron than semaglutide, but the semaglutide used in the study was the Rybelsus lower dose (7 or 14 mg). Orforglipron users were more likely than semaglutide users to discontinue the drug because of side effects.
Research: Early Stage Lung Cancer Surgery Safe for Many Patients Over 80
Researchers at the Icahn School of Medicine and the Mount Sinai Tisch Cancer Center in New York found that adults aged 80 and older with early-stage lung cancer can safely undergo surgery and achieve outcomes comparable to younger patients, challenging longstanding assumptions about age and cancer treatment. “As our population ages, more patients over 80 are being diagnosed with early-stage lung cancer, yet they are often not considered for surgery,” said lead researcher Raja M. Flores, MD, Chair of the Department of Thoracic Surgery at Mount Sinai Health System. “Our findings show that when patients are carefully selected based on their overall health, not just their age, they can tolerate surgery well and experience excellent long-term outcomes.”
The study followed 884 patients with early-stage lung cancer, including 114 people who were age 80 or older, finding that older patients lived just as long as younger patients after surgery. While some older patients had more complications right after surgery, most patients in both groups felt better over time, and their quality of life improved within a year. The findings were published in The Lancet Regional Health – Americas.
Mount Sinai Press Release: Mount-sinai-study-finds-lung-cancer-surgery-safe-for-many-patients-over-80
Supplement Tips: Fish Oil Capsules Safety
Consumer Reports recently tested 20 popular fish oil supplements for safety and quality standards, including testing for heavy metals and contaminants. Sixteen of them passed, but these four 1000 mg “softgels” products did not meet label claims for omega-3 fatty acid content or showed signs of rancidity: Kirkland Signature (Costco) fish oil softgels, Nature Made Burp-Less Fish Oil Softgels, California Gold Nutrition (Kids Omega-3 Fish Oil Softgels, Strawberry), Qunol Ultra Omega-3 Mini Softgels.
Read the complete report for more insights into fish oil supplements.
Consumer Reports: “What’s in Your Fish Oil Pill?” (April 1, 2026)
Did you miss the last edition of the newsletter? Check it out here:

