Nationwide Recall of Alcon, iVizia, Optane Eye Gel Drops; Furosemide, Pantoprazole, Prescription-Strength Fish Oil; New and Experimental Drug Approvals; Aspirin and Cancer Study
New Treatment for Agitation Associated with Dementia due to Alzheimer’s: Auvelity
The FDA granted expanded approval for Axsome Therapeutic’s Auvelity extended release tablets (dextromethorphan hydrobromide and bupropion hydrochloride) for the treatment of agitation associated with dementia due to Alzheimer’s disease. The only other drug approved for agitation associated with dementia due to Alzheimer’s is Otsuka’s Rexulti (Brexpiprazole), an antipsychotic approved in 2023 for that condition. Rexulti has a boxed warning that elderly patients with dementia-related psychosis treated with antipsychotic drugs are at increased risk of death; it also has more severe side effects overall, so this new approval seems to offer a safer alternative.
FDA initially approved Auvelity in 2022 to treat major depressive disorder in adults.
FDA Press Release: FDA-approves-first-non-antipsychotic-drug-treat-agitation-associated-dementia
Safe to Proceed: Pancreatic Cancer Experimental Treatment Daraxonrasib
The FDA sent a “safe to proceed” letter to Revolution Medicines, allowing access to an experimental pancreatic cancer treatment for patients not involved in current clinical trials. The investigational treatment, Daraxonrasib, is a pill taken daily by patients with previously treated metastatic pancreatic ductal adenocarcinoma (PDAC). Under the “Expanded Access Program” rules, requests for treatment must be submitted to Revolution Health by physicians licensed in the U.S. on behalf of eligible patients. Eligible patients are those with previously treated metastatic PDAC who are not eligible for participation in a Revolution Medicines-sponsored clinical trial or are unable to access an appropriate clinical trial and have no satisfactory alternative treatment options. Revolution Medicines will provide the treatment free of charge under this program.
Former Nebraska senator Ben Sasse is a participant in the Phase 3 clinical trial and has credited Daraxonrasib for extending his life and improving his symptoms. Sasse was diagnosed with stage 4 metastatic pancreatic cancer late last year and given a survival prognosis of only about three months. Revolution Medicine reports that its Phase 3 trial showed that the drug almost doubled the typical length of survival and slashed the risk of death by 60% versus chemotherapy. The sponsor plans to seek expedited Food and Drug Administration approval using a Commissioner’s National Priority Voucher, which may mean the drug will be approved within weeks of submission of the application.
Revolution Medicine Expanded Access Policy.
FDA Press Release: FDA permits-expanded-access-investigational-pancreatic-cancer-drug
CNBC: Pancreatic-cancer-drug-daraxonrasib-from-revolution-medicines-succeeds-in-trial.html
Gene Therapy for Deafness in Children: Otarmeni
The FDA approved Regeneron’s Otarmeni, the first gene therapy for otoferlin deafness, a rare type of genetic hearing loss accounting for about 2 percent to 8 percent of people with inherited deafness About 50 babies are born each year with the condition.
The FDA based its approval on results of a clinical trial in which 20 children, aged 10 months to 16 years with otoferlin gene-associated severe-to-profound and profound hearing loss. The treatment improved hearing in 80 percent of the participants within a year of their surgery, and 42 percent of them could hear so well they could detect whispers. The approval was issued 61 days after BLA filing, marking the sixth approval under the Commissioner’s National Priority Voucher (CNPV) pilot program and the first gene therapy product approved under the program.
In gene therapy, a problematic gene is replaced with a working version. To deliver Otarmeni, doctors make a small incision and deliver the new gene into the cochlea, a snail-shaped organ in the inner ear. Regeneron says it plans to provide the therapy for free to patients in the United States.
FDA Press Release: FDA-approves-first-ever-gene-therapy-treatment-genetic-hearing-loss-under-national-priority-voucher
Smithsonian Magazine: The FDA-approves-the-first-ever-gene-therapy-for-deafness-which-aims-to-restore-hearing-in-kids-with-a-rare-inherited-condition-180988635/
Juvenile Ankylosing Spondylitis (JAS) and Hidradenitis Suppurativa in Adolescents: Cosentyx
Novartis’ psoriasis drug Cosentyx received expanded approval for treating juvenile ankylosing spondylitis (JAS), a type of arthritis of the spine, in pediatric patients age 12 and up. Cosentyx also received approval to treat patients age 12 and up with moderate to severe hidradenitis suppurativa (HS), a painful, chronic skin disease also called acne inversa. HS usually starts after puberty, usually in the teens or twenties, and is more common in women. The approvals were based on safety and efficacy data from well-controlled trials of adults with ankylosing spondylitis. The medication, delivered monthly (after initiation doses) by subcutaneous injection, was originally approved in 2015.
Revised Safety Information and Label Changes
Read Cosentyx Patient Reviews for Treating Ankylosing Spondylitis
Novartis Press Release
Unexplained Spontaneous Hives (“uncontrolled chronic urticaria”) in Young Children: Dupixent
Sanofi and Regeneron’s eczema drug Dupixent (dupilumab) received expanded approval for treating uncontrolled chronic spontaneous urticaria (CSU) in children age 2 and up. The approval was based on a trial of 18 patients age 2 to 11 years of age who received DUPIXENT based on weight, with no new adverse reactions identified. Previously, Dupixent was approved for CSU in patients 12 years old and up. Dupixent helps control the inflammation that leads to hives. Given as a subcutaneous injection every two weeks, Dupixent was originally approved in 2017 for atopic dermatitis (eczema). It now is approved for nine different conditions including asthma, allergic fungal rhinosinusitis, chronic obstructive pulmonary disease (COPD), chronic rhinosinusitis with nasal polyps.
Read more about chronic spontaneous urticaria (CSU), a condition in which hives break out frequently without an apparent reason, at the Asthma and Allergy Foundation of America.
Revised Dupixent Label and Approval Letter for New CSU indication
Strengthened Warnings about Inflamed Eyes with Dupixent
Blepharitis (inflammation of the eye lid), primarily in eczema patients, has been reported by patients taking Dupixent. The label now warns that some patients reported varying degrees of transient or ongoing visual impairment including blindness associated with conjunctivitis, keratitis, or blepharitis leading to discontinuation of Dupixent and/or surgical intervention. Dupixent should be used with caution in patients with significant dry eye disease, history of significant lid abnormalities/surgeries, or history of tear duct surgery. Patients are advised to promptly consult their healthcare provider if new onset or worsening eye symptoms develop.
Label Change (April 16, 2026) for Eye Condition Warnings
RECALLS:
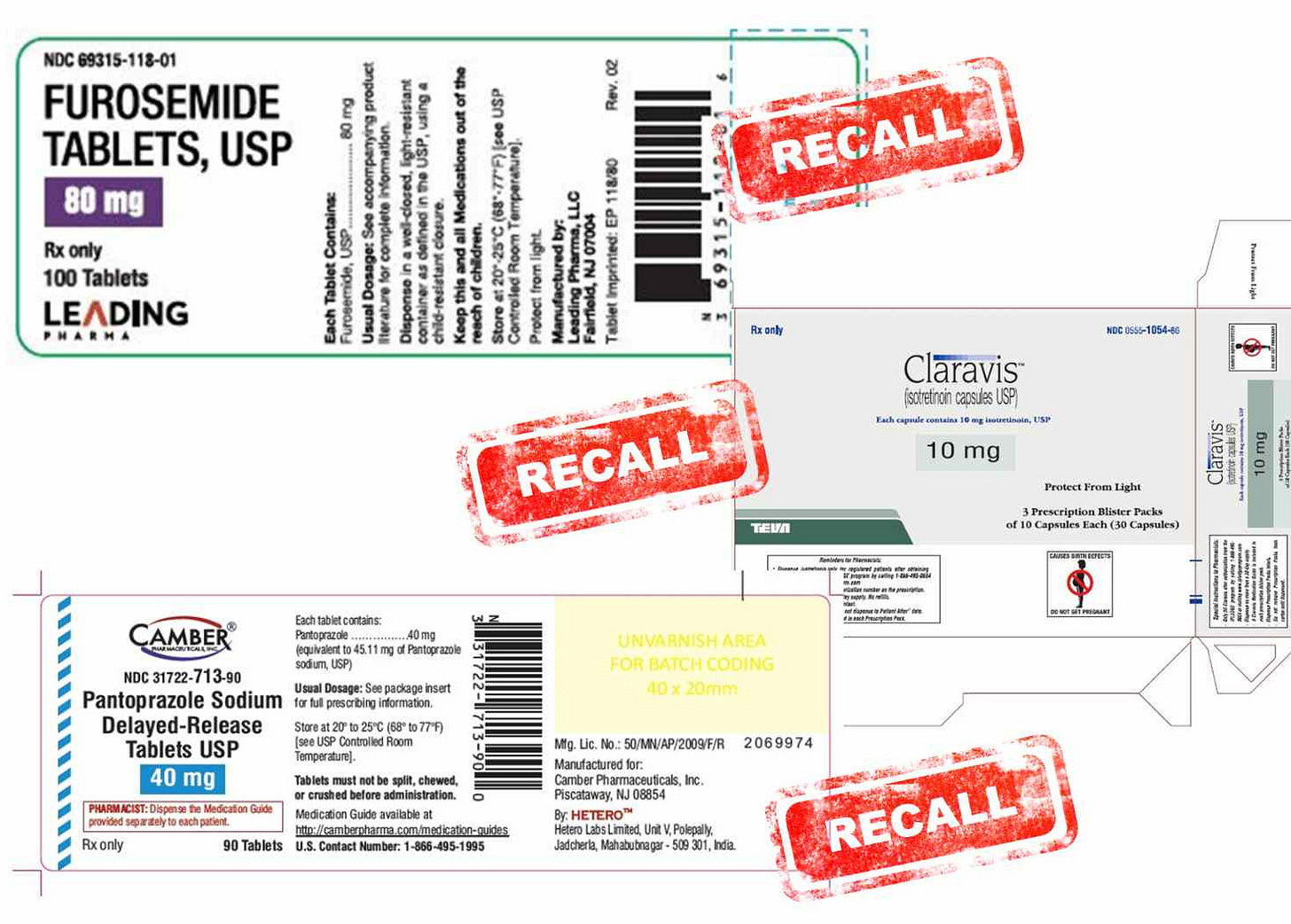
Furosemide Tablets Recall by Leading Pharma
Leading Pharma is recalling 9,3840 bottles of 80 mg furosemide tablets (100-count) because of the presence of nitrosamine, a cancer-causing impurity, above the FDA-allowed limit. Furosemide (Lasix brand name) is a diuretic that reduces water in the body and often is used for heart failure, high blood pressure, and edema. The NDC number, which should appear on your medication bottle or label, is 69315-118-01. Check the announcement for specific lot numbers and expiration dates.
FDA Recall Enforcement Report
Pantoprazole Tablets Recall by Hetero Labs
Hetero Labs is recalling 4,740 1000-count bottles of 40mg pantoprazole tablets manufactured in India for Camber Pharmaceuticals due to customer complaints about tablet discoloration. Pantoprazole (generic for Protonix) is a proton pump inhibitor (PPI) that treats acid reflux, gastritis, or other gastrointestinal problems. The image below is for 90-count bottles; the recalled 1000-count bottles were likely distributed to pharmacies and later put into smaller containers. The NDC number, which should appear on your medication bottle or label, is 31722-713-10. Check the announcement for specific lot numbers and expiration dates.
FDA Recall Enforcement Report
Protonix Patient Reviews at Ask a Patient and Pantoprazole Patient Reviews
Claravis Capsules Recall by Teva
Teva is recalling 5,101 cartons of 100-count blister packs of 10 mg Claravis (isotretinoin) due to a manufacturing quality problem resulting in “out of specification for specific impurity tretinoin.” This could affect product stability and effectiveness. Claravis is a branded generic and treats severe cystic acne. Check the announcement for specific lot numbers and expiration dates.
FDA Recall Enforcement Report
Fun Pharma Fact
Did you know that there are three types of generic drugs? Traditional generics are the most common. Authorized generics are produced to be carbon copies of the brand name version, but go by the generic name, and are not always available. “Branded generics,” like Teva’s Claravis, are generics labeled with a “brand” name. Find out how these types compare with respect to when they can be sold, whether they can be advertised, cost, examples, and more.
Ask a Patient: “Generic Drug Types Explained: Unbranded, Authorized, and Branded”
Omega-3-Acid Ethyl Esters Capsules Recall by Major / Harvard
Harvard Drug Group is recalling 2,238 cartons of prescription-strength fish oil, Omega-3-acid ethyl esters capsule 1 g. due to the capsules failing stability testing. Packaged in 10-unit dose blister cards, the capsules were packaged and distributed by Major. Omega-3 acid ethyl esters are a type of fat found in fish oil and used along with diet and exercise to help lower levels triglyceride, a type of blood fat. It may also raise “good” cholesterol (HDL). The product NDC number for this recall is 0904-7495-06. Check the announcement for lot numbers and expiration dates.
FDA Recall Enforcement Report

Check out reviews for Lovaza or add your review for a generic Omega 3 Acid Ethyl Esters Rating Form
There have been multiple eye drops recalls this year. Today we feature four recent over-the-counter eye drop recalls, with sample product images presented below.
GenTeal Tears and Systane Lubricant Night Eye Gel Recall by Alcon Labs
Alcon Laboratories is recalling an unspecified number of bottles of GenTeal Tears Lubricant Eye Gel Drops (10g or 0.34 Fl oz) due to a lack of assurance of sterility. The NDC number for this product is 0065-8064-01. Check the FDA recall report for specific lot numbers and expiration dates.
As part of the same recall, Alcon is also recalling an unspecified number of bottles of Systane Lubricant Eye Gel, Night Gel, Sterile, 10g (0.35 oz) due to a lack of assurance of sterility. The drops were processed in France for Alcon and the NDC number for this product is 0065-0474-01. Check the FDA recall report for specific lot numbers and expiration dates.
FDA Recall Enforcement Report
iVIZIA Lubricant Eye Drops Recall by Thea Pharma
Thea Pharma is recalling an unspecified number (five lots) of 0.33 Fl oz (10 mL) bottles of iVIZIA, Sterile Lubricant Eye Drops (Povidone 0.5%) due to a lack of assurance of sterility that was found by the FDA during an inspection of the manufacturer. The product was made in France. FDA Recall Enforcement Report
Although this recall was issued at the retail (not consumer) level, iVIZIA posted a detailed recall announcement on its website with lot numbers, product photo, expiration dates, and instructions to consumers: iVIZIA Recall Announcement
Optase Dry Eye Intense Drops Recall by Scope Health
Scope Health is recalling an unspecified number of Optase Dry Eye Intense Drops (Glycerin 0.2%), packaged in 0.33 fl oz bottles due to a lack of assurance of sterility. The NDC number is 72972-002-01 Check the FDA Recall Enforcement Report for specific lot numbers.
FDA Recall Enforcement Report

Medication Tips: Aspirin and Potential Health Benefits
Taking daily low-dose aspirin can be helpful for prevention of cardiovascular disease or stroke for certain people (under a doctor’s guidance). But with its anti-inflammatory properties, could it also help reduce cancer risk? A Newcastle University study of people with Lynch syndrome (gene mutation that increases your chance of developing colon cancer), effectively halved participants’ risk of getting colon cancer for those taking daily low-dose aspirin for two years. Similar studies have been done or are underway, including one with 11,000 participants who’ve had colorectal, breast, gastroesophageal, or prostate cancer in the UK, Ireland and India.
BBC: “Aspirin can reduce the risk of cancer - and we’re starting to understand why”(April 2026)
Last week, Dr. Lucy McBride, writer of “Are You Okay?” answered a reader-submitted question on the topic:
“I just read that aspirin may help prevent certain cancers and slow their spread. Is this true? I’ve heard about all kinds of benefits for Aspirin and the conversation keeps changing. Should I be taking Aspirin every day or are there risks I am missing?”
Read her answer: Aspirin and Cancer Risk
Did you miss the last newsletter edition? Check it out here: