Estradiol and Testosterone Recalls; Metoclopramide Warnings; Approvals for Pancreatic and Ovarian Cancer; Landmark PBM Reform; Pharma TV Commercials Flagged; Psychiatric Drugs and More
Recall of Estradiol and Testosterone Pellets from U.S. Compounding Outsourcing Pharmacy
Asteria Health, a licensed compounding outsourcing pharmacy in Birmingham Alabama, is recalling more than 118,000 pellets of estradiol ranging in strength from 6 mg to 37.5 mg along with more than 600,000 testosterone pellets and testosterone-anastrozole combination pellets ranging in strength from 12.5 mg to 200 mg due to potential metal particle contamination. These pellets are administered subcutaneously (under the skin). Testosterone pellets treat testosterone deficiency or hypogonadism. Estradiol pellets, also a “bioidentical hormone replacement therapy, ” are often used for menopause symptoms. Asteria is part of Biote Health, a provider of hormone imbalance therapies for men and women.
FDA Recall Enforcement Report
Read more about hormone pellet therapy at Medical News Today: “Hormone Pellet Therapy for Menopause”
TV Commercials Flagged for False and Misleading Advertising
Sponsors of the following drugs recently received letters from the FDA warning that promotional communications, including direct-to-consumer advertisements for the drugs are “false or misleading” because they make false claims about clinical trials results and drug efficacy, omit important information, and/or present visuals that are misleading. These letters are sent from the FDA Office of Prescription Drug Promotion (OPDP).
Tremfya (guselkumab) injection for plaque psoriasis: OPDP Letter
Neffy (epinephrine) nasal spray for emergency allergic reactions including anaphylaxis: OPDP Letter
Here is an example of concern cited by the FDA: actors in the commercial are shown pulling a bottle of Neffy out of a purse or other bag in order to demonstrate how compact and convenient the medication is. In reality, the patient guide clearly states that the medication must be kept in its blister pack until it is time to use the medication.Wegovy (semaglutide) for weight loss: OPDP Letter
Vonjo (pacritinib) for a rare bone marrow cancer called myelofibrosis: OPDP Letter
Check here for a list of 103 FDA “untitled” letters for pharmaceutical products from the Office of Prescription Drug Promotion going back to 2018. The list can be sorted by product name, company name, and date of letter.
New Drug Approvals Will Require a Single Clinical Trial
FDA Commissioner Martin Makary and Vinay Prasad, director of the Center for Biologics Evaluation and Research (CBER) announced (in the New England Journal of Medicine) the FDA’s plan to require only one clinical trial (instead of two) to serve as the basis for approving new drugs. Although the two-trial requirement has been in effect since the 1960s, new types of approval paths, such as those for rare diseases, have gained at least temporary approval on the basis of just one trial.
Previous FDA Commissioner Janet Woodcock, who supports the change, said that more than half of drug approvals happening now are already based on just one trial. Previous FDA Commissioner Robert Califf also expressed his support of the new policy, stating that “the FDA will still retain discretion to require additional studies when warranted,” but says more detail is needed. Other former top FDA officials who spoke to RAPS Focus raised concerns about how the policy was developed and announced.
RAPS Focus: “Experts React to FDA Shift to Single Pivotal Trial for Most Drugs”
New England Journal of Medicine: “One Pivotal Trial, the New Default Option for FDA Approval — Ending the Two-Trial Dogma” (requires subscription to view)
First Pancreatic Cancer Device Treatment
The FDA approved Novocure’s Optune Pax as the first of its kind device treatment for adults with locally advanced pancreatic cancer, to be used in conjunction with chemotherapy. The portable non-invasive device delivers alternating electrical fields, known as tumor treating fields (TTFields), to the abdomen. TTFields work by physically disrupting the rapid cell division that is characteristic of cancer cells, while minimizing damage to healthy tissue. Oncologist Daniel Flora notes that although this is not a breakthrough treatment (only eight weeks of overall survival benefit), it is worth considering for a cancer type that has few available treatment options.
Novocure also makes products that work in a similar way for brain cancer (approved in 2015) and lung cancer (approved in 2024).
FDA Press Release: FDA approves-first-its-kind-device-treat-pancreatic-cancer
Fierce Biotech: “Novocure gets FDA nod for tumor-treating electric fields targeting pancreatic cancer

Keytruda Approved for Ovarian Cancer
The top-selling drug in the world with over $31 billion in sales, Keytruda was just approved for an additional cancer indication: ovarian cancer. It is also now available for subcutaneous injection as Keytruda QLEX. This chart from Senthil Ramanathan Ph.D. shows rankings of oncology drugs by worldwide revenue in billions.
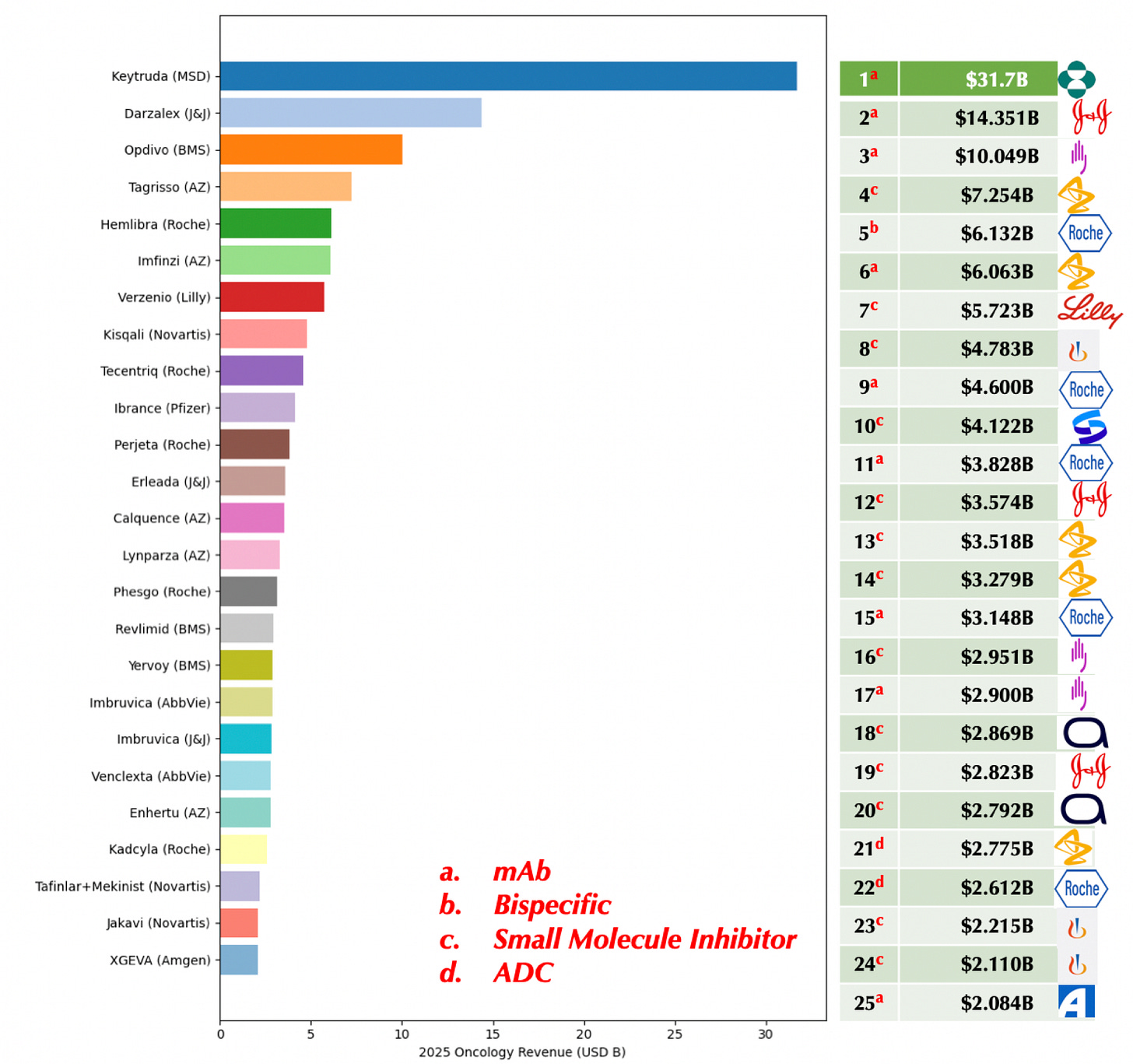
FDA Press Release: “FDA approves pembrolizumab with paclitaxel for platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma”
If you have used Keytruda as a treatment for any illness, did it help? Please take a moment to add your experience (and/or rate it on a scale of 1 to 5).
Warnings Strengthened about TD Risk for Metoclopramide-Containing Drugs
Drug labels for metoclopramide-containing drugs including Reglan (metoclopramide hydrochloride tablets and metoclopramide hydrochloride injection) and Gimoti (metoclopramide hydrochloride nasal spray) have been revised to strengthen the warnings about the risk of developing tardive dyskinesia (TD), a movement disorder that is more likely to happen with long-term use. Metoclopramide, which was approved in 1979, added a black box warning about the risk of TD in 2009.
Reglan tablets are approved for diabetic gastroparesis (a delay in the movement of stomach contents) and short-term treatment of gastroesophageal reflux (GERD) in patients who don’t respond to conventional therapies. In its I.V. form, it is approved for the prevention of nausea for chemotherapy and after surgery. Gimoti nasal spray is approved for treating diabetic gastroparesis. Read the safety label update and corresponding revised labels for all the changes, which also affect generic versions of the medications.
Safety Label Update for Reglan injection (currently available as generic only)
Safety Label Update for Reglan tablets
Safety Label Update for Gimoti nasal spray
The Columbian: “Vancouver-woman-sues-legacy-health-kaiser-permanente-and-two-physicians-after-developing-involuntary-movement-disorder”
This article describes the experience of a woman who had taken Reglan for more than two years as a treatment for gastroparesis. She first experienced restless legs syndrome then later an irreversible form of tardive dyskinesia; the drug continued to be prescribed even when switching doctors, and she was unaware of the possible connection of her symptoms with the drug. Thousands have filed lawsuits or complaints against Reglan and other brands of metoclopramide because of these effects.
Reglan FDA Adverse Event Summary by Ask a Patient show muscle-related disorders (tardive dyskinesia, extrapyramidal disorder, and muscle rigidity) as the top three reported adverse effects for the over 19,000 reports including Reglan between 2004 and 2025. Among the 566 voluntary reports filed directly through the Medwatch system, tremor was the 7th most commonly reported adverse effect.
For Gimoti, tremor is the 7th most commonly reported adverse effect on the report with 33 reports of tremor within the 539 total reports received for Gimoti so far.
View or add patient reviews for brand name Reglan or generic or Gimoti at Ask a Patient.
Note that the FAERS database (FDA Adverse Event Reporting System) consist mostly of company-submitted reports, which are mandatory. Reports submitted by individuals (consumers, doctors, or pharmacists) are voluntary reports and are submitted through the “MedWatch” database and represent just a small (less than 5%) of all reports in the FAERS database. Access one-page highlights of FAERS and MedWatch reports for thousands of drugs at Ask a Patient website.
Poor Report Card for Psychiatric Drugs Innovation
A University of Chicago/Stanford study found that psychiatric drug development lags behind other medical fields in terms of novelty and clinical impact. Researchers investigated clinical utility, innovation, and other measures of helpfulness as rated by various international organizations and regulatory bodies for 22 psychiatric drugs approved between 2012 and 2024.
Of the 22 drugs, 7 (31.8%) were categorized as first-in-class, 2 (9.1%) were considered an advance-in-class, and 13 (59.1%) were considered addition to-class. Three drugs (13.6%) received FDA priority review, 1 (4.5%) was designated as an orphan drug, and 0 were included on the WHO’s Model List of Essential Medicines (see below for the list of WHO-included psychiatric drugs). For clinical utility, none of the drugs with data available data received a rating of “clinically helpful,” and 3/22 (13.6%) were rated “clinically not helpful. The study was published in the Journal of Clinical Psychiatry.
Journal of Clinical Psychiatry: “Innovation in Psychiatric Drug Development: A Quantitative Analysis of FDA-Approved Psychiatric Drugs, 2012–2024”
Ask a Patient: “Changes to the WHO 2025 Model List of Essential Medicines for Mental and Behavioral Disorders”
Landmark PBM Reform Legislation and FTC Settlement: Good News for Consumers and Community Pharmacies
New federal laws combined with stepped-up Federal Trade Commission enforcement (such as the recent settlement with Cigna) of the PBM industry could mark 2026 as “the year of PBM reform.” Congress and the FTC are putting an end to the tactics used by PBMs to continue their rebate-hoarding practices, which included setting up skeleton-crew revenue receiving operations (called “Group Purchasing Organizations”) in tax-sheltered countries like Switzerland. All of the required changes, plus changes still to come from unsettled FTC action against CVS and United Healthcare, are good news for community pharmacies (independent pharmacies) and signal lower drug prices for consumers.
Investigative news organization Hunterbrook Media spent a year investigating the mysterious group purchasing organizations run behind the scenes by the big three PBMs (CVS, United Health, and Cigna). Hunterbrook’s investigative report “The Hidden Flow of Drug Money” was entered into the Congressional record in a recent hearing where the big three health insurance executives were questioned by lawmakers. Click on the link in the caption below to access the page with the interactive graphic that clearly explains the findings. “Drug manufacturers pay billions in discounts, rebates and fees to pharmacy benefit managers (PBMs). But where does that money actually go? Follow the flow.”
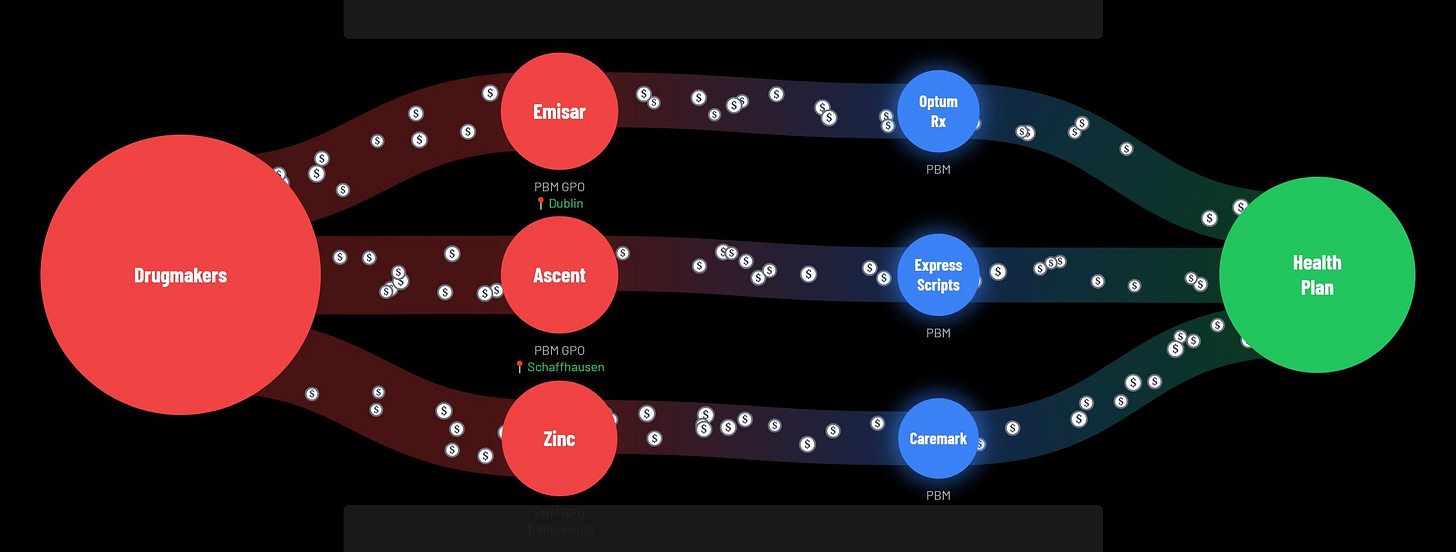
From Hunterbrook: “Our multinational investigation reveals how CVS, United Health, and Cigna created new subsidiaries to divert billions of dollars from health plans and patients. All three tried to keep it secret. None answered repeated questions. CVS sued to stop evidence getting out. Cigna called the police on a reporter.”
“The takeaway: PBM GPOs appear to generate astronomical revenue with skeleton staff — making Zinc (CVS), Emisar (United Health), and Ascent (Cigna) among the world’s most lucrative enterprises on paper, even though they appear barely to exist in the real world…”
Read about the recent FTC settlement with Cigna/Express Scripts PBM from HealthCare Uncovered: “In FTC Settlement, Cigna Agrees to Change Some PBM Business Practices, Charge Customers Less for Insulin”
Read about new legislation that has been signed into law from KFF News: “What to Know About Pharmacy-benefit-managers-pbms-and-federal-efforts-at-regulation”
The National Community Pharmacists Association (NCPA), which represents independent (not chain) pharmacies, applauds the recent PBM reform. “For years, our members and we have been telling anyone who will listen — and worked to convince others who wouldn’t listen — about the PBM-insurer conglomerates gobbling up market share, driving up drug costs, crushing small-business pharmacies, and making it more difficult for patients to receive the care they need. We’ve been warning that unless action is taken, more pharmacies will close, and more pharmacy deserts will grow. Unfortunately, as time passed, we were proven correct and finally, there is action to help reverse these trends.”
NCPA Cheers Trump Signs First Major PBM Reform in Decades
They also applaud the FTC action against Express Scripts:
NCPA Press Release: Boom! FTC Squeezes Concessions from Cigna’s Express Scripts
Did you miss the last edition from February 8? Find it here:


